IMT Application 2026
Dear Friend,
Hope you’ve had a good week. Mine has been filled with the usual mix of ward work, bleeps, and inbox firefighting — but also an odd sense of déjà vu. Every year around this time, conversations among juniors shift. The topic changes, the rota doesn’t. This week it’s been all about IMT applications, and how different things will look in 2026.
And if I’m honest, those conversations have felt heavier than usual.
Because each year, the process gets more competitive.
And each year, more and more brilliant colleagues — genuinely excellent doctors — don’t even score an interview.
Not because they’re poor clinicians.
What last year showed us
The 2025 shortlisting score made something painfully clear: to secure an IMT interview, clinical skill wasn’t the deciding factor.
It was points.
Publications, presentations, audits, teaching programmes — essentially a portfolio of achievements that often say more about strategy than day-to-day doctoring.
Many great clinicians found themselves below the cut-off simply because they didn’t have a string of PubMed IDs or national presentations. And that stings, especially for those who spent more time looking after patients than chasing posters.
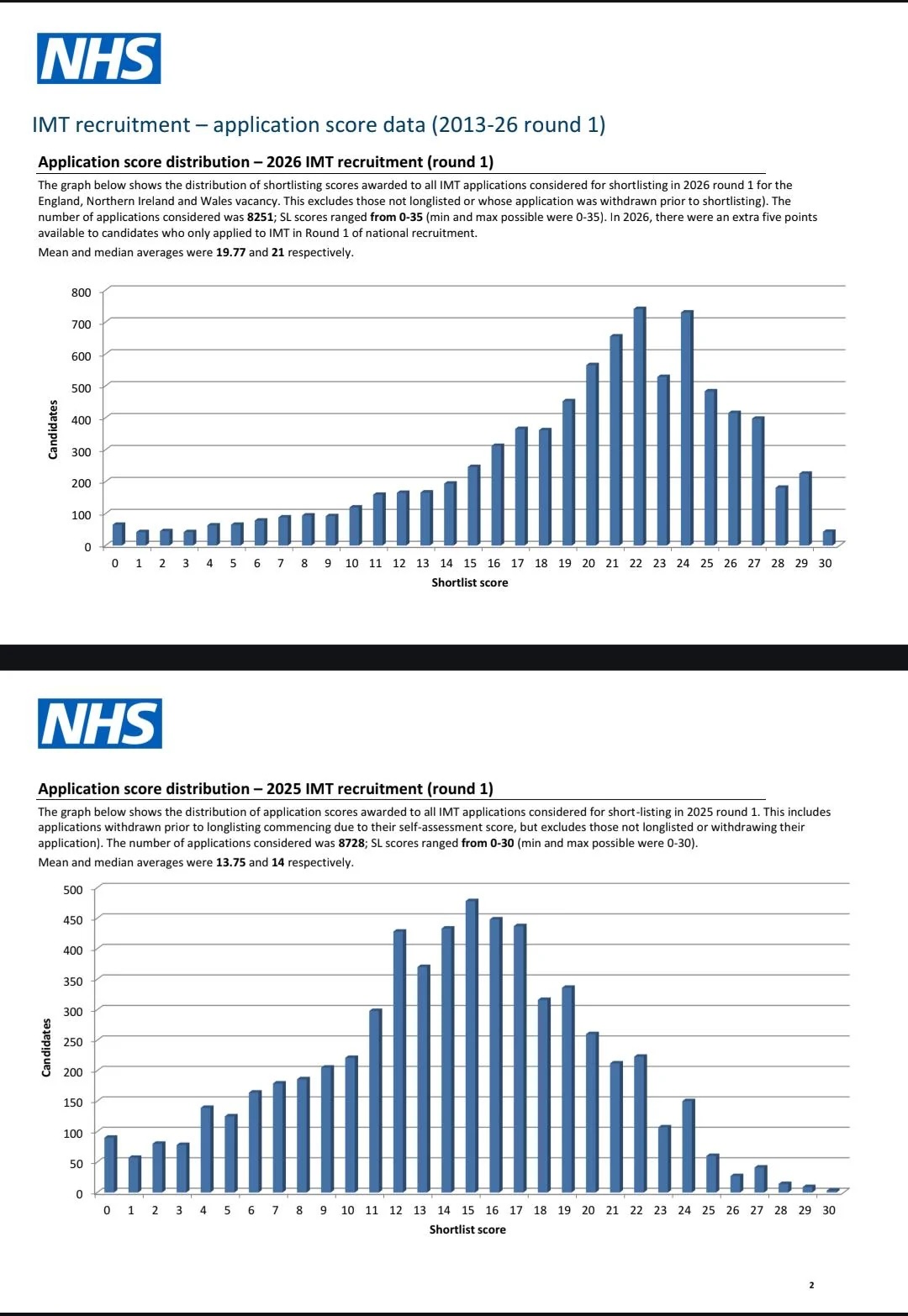
The numbers don’t lie
Last year, over 8,800 doctors applied for roughly 1,700 IMT places.
A competition ratio that would make even neurosurgery blush.
So, did they create more IMT posts?
Of course not.
Did they prioritise UK graduates, who spend years training in a system that now cannot absorb them?
No.
Instead, they introduced a new “solution”:
an extra 5 points for candidates who apply only to IMT.
On paper, the idea is simple — discourage people from applying broadly to IMT, GP, psych, paediatrics all at once. Reduce the applicant pool. Improve the ratios.
In reality?
It just made things more strategic.
If you apply to multiple specialties, your score is now automatically 5 points lower. And unless you’ve absolutely maxed out every other scoring domain, that difference is enough to cost you an interview.
And can we blame people for maximising their chances of getting a job?
In this market? Absolutely not.
But what about the doctors who commit solely to IMT — and still don’t get in?
They don’t have a backup. They don’t suddenly gain extra posts to apply for. They simply… fall through the cracks.
So… did it work?
The early data is telling:
The average applicant score has gone up — across the board. (see photo above).
The number of spaces available is unchanged.
In other words:
The new rule didn’t reduce applicant numbers.
It just raised the bar even higher for everyone.
And the people who suffer most are the ones who didn’t know how “strategic” they needed to be.
Summary
I don’t pretend to have the answers.
But I do know this: we’re long past the point where passion, skill, and clinical competence are enough. And that’s a hard thing to swallow — for trainees, for mentors, for anyone who entered medicine to care rather than score.
If you’re applying this year, my heart goes out to you.
If you’re supporting someone who is, remind them that their worth is not a number out of 60.
And if you’re watching from the sidelines — well, welcome to the annual IMT Hunger Games. May the odds, and the publications, be ever in your favour.
Drug of the week
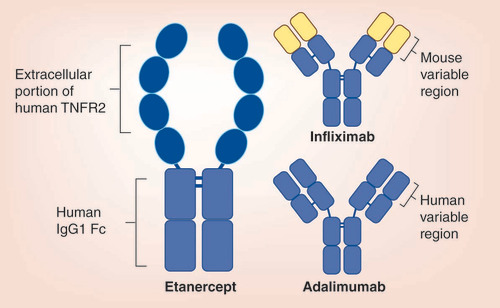
Etanercept
Etanercept is a biologic DMARD used for conditions like rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis.
It’s a soluble TNF receptor fusion protein that binds circulating TNF-α, preventing it from interacting with its natural cell-surface receptors.
By blocking TNF-α signaling, it quickly reduces inflammation and helps slow joint and tissue damage.
It’s given by subcutaneous injection, usually after traditional DMARDs aren’t enough.
Common side effects include injection-site reactions, headache, and mild respiratory symptoms.
Because it suppresses TNF-α, it increases infection risk—especially TB—so patients are screened before starting.
Avoid use in active infections; use caution in heart failure and demyelinating disease.
Etanercept has a relatively low risk of antibody formation, helping maintain stable long-term response.
A Brain Teaser
A new monoclonal antibody drug is being developed to treat severe atopic dermatitis. The research group wants to compare this drug to placebo in 20 patients with severe atopic dermatitis. This is so the researchers can collect some preliminary data regarding the efficacy and optimal dosing of this antibody.
What phase of the clinical trial is the drug entering?
A: Phase 1
B: Phase 2
C: Phase 3
D: Phase 4
E: Phase 5
Answers
The answer is B – phase 2.
Phase 2 is correct as this phase involves small-scale drug testing in a target patient population, which in this case is patients with severe atopic dermatitis. Phase 2 may sometimes be subdivided into 2a and 2b, where the optimal dosing and efficacy of the drug are assessed respectively. Researchers will then use this evidence to help attract investment for larger phase 3 trials designed to determine the overall effectiveness of the treatment.
Phase 1 is incorrect as phase 1 clinical trials are conducted on healthy volunteers not target patient populations. This is to establish pharmacokinetics, pharmacodynamics and adverse effects of a drug before larger studies are conducted.
Phase 3 is incorrect as phase 3 clinical trials involve a full randomised control trial with larger sample sizes (typically 100 to 100,000) than the group of 20 outlined in this question. This is often used to compare the new drug to the existing standard treatment, or in some cases to a placebo.
Phase 4 is incorrect as phase 4 clinical trials only happen after a drug or device has been approved and brought to market. The purpose of phase 4 is to monitor for longer-term effectiveness and adverse events, sometimes referred to as post-marketing surveillance. However, note that this phase is not always performed.
Phase 5 is incorrect. There is no fifth phase in clinical trials.



