How much do patients understand?
Dear Friend,
This week, I found myself back in student mode, working on an essay for my statistics module as part of my oncology masters. It started off as expected — papers, methods, outcomes — but one study in particular stayed with me.
What I Was Reading
The paper, by Tagai et al., looked at how well patients with prostate cancer understand the Gleason score. What it found was simple, but quite striking: around 50% of patients did not have an adequate understanding of what their Gleason score actually meant.
This isn’t a niche concept. It’s something we use routinely — in clinic, in letters, in discussions about prognosis and treatment. And yet, for many patients, it isn’t landing.
In simple terms, the Gleason score is a way of grading prostate cancer based on how abnormal the cells look under a microscope. Two patterns are identified, each scored from 1 to 5, and then added together. So a result might be written as 3 + 4 = 7.
The higher the score, the more aggressive the cancer is likely to be.
It’s one of the main tools we use to guide management — from active surveillance to surgery or radiotherapy. From our perspective, it’s fairly straightforward. But that paper suggests something different: clarity for us does not mean clarity for patients.
What Does the Evidence Show More Broadly?
That paper led me to a broader question.
How much do patients actually understand what we tell them?
There’s quite a bit of research on this, and the findings are fairly consistent.
Patients often report that they understand the information given to them — but when their understanding is tested, it’s frequently incomplete or incorrect.
In some cases, this gap is quite large. One study comparing clinician and patient perceptions found that while 75% of doctors believed they had communicated effectively, only 21% of patients felt satisfied with that communication.
Even when looking specifically at treatment plans, understanding is variable. In one inpatient study, only around two-thirds of patients had a clear idea of their management plan.
And when it comes to medication, the picture is similar. Health literacy studies suggest that a significant proportion of patients struggle to understand written medication information, with estimates that up to 60% have limited health literacy.
Taken together, the pattern is fairly clear. We often think we’ve explained things well. Patients often think they’ve understood. But the reality sits somewhere in between.

Why This Matters
Understanding isn’t just a nice extra. It underpins everything — consent, adherence, decision-making.
If a patient doesn’t fully understand their diagnosis or treatment, it becomes much harder for them to engage with it in a meaningful way. And the difficulty is that misunderstanding is often invisible.
Patients may not ask questions. They may not want to appear confused. They may feel they should understand. So we move on, assuming the message has landed.
What Can We Do Better?
There are small changes that seem to make a real difference. Avoiding jargon is the obvious starting point. Not just technical language, but the everyday medical shorthand we don’t notice we’re using.
Checking understanding is probably even more important. Asking patients to explain things back in their own words — the “teach-back” method — is a simple way of seeing whether what we said actually made sense. And then there’s how we document things.
Clinic letters are increasingly read by patients themselves. Writing these in clear, plain language matters. They often become the reference point patients return to after the consultation. None of this is complicated. But it does require us to be a bit more deliberate.
Final Thoughts
That paper was framed around the Gleason score. But it didn’t really feel like it was about prostate cancer. It felt like a reminder of something more basic — that understanding isn’t guaranteed just because we’ve explained something.
Most of the time, we assume it’s there. And most of the time, we probably overestimate it.
Drug of the week
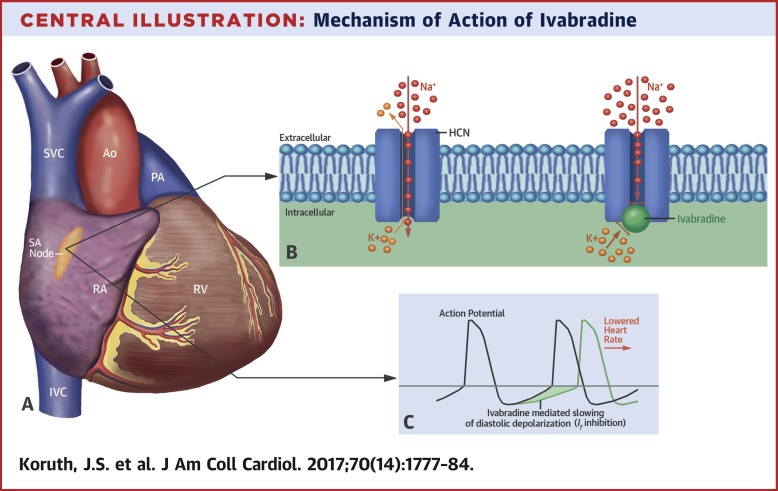
Ivabradine
Ivabradine is a selective sinus node inhibitor used in chronic heart failure and stable angina.
It works by inhibiting the If (“funny”) current in the sinoatrial node, reducing heart rate without affecting myocardial contractility or blood pressure.
It acts specifically on pacemaker activity and has no effect on cardiac conduction or ventricular repolarisation.
It is used in patients with heart failure with reduced ejection fraction who remain symptomatic despite optimal therapy, and in those unable to tolerate beta-blockers.
Common side effects include bradycardia, headache, and visual disturbances, particularly transient luminous phenomena (phosphenes).
Atrial fibrillation can occur and patients should be monitored for new-onset arrhythmias.
Ivabradine is contraindicated in patients with significant bradycardia, acute decompensated heart failure, and in combination with strong CYP3A4 inhibitors.
A Brain Teaser
A 58-year-old man attends his GP for a routine check-up. He feels well and has no specific complaints. He has a history of type 2 diabetes mellitus and hyperlipidaemia. His medications include metformin and atorvastatin.
On examination, his blood pressure is 152/96 mmHg. A repeat reading taken 10 minutes later is 148/94 mmHg. His BMI is 29 kg/m². Cardiovascular and respiratory examinations are unremarkable.
Blood tests show:
- HbA1c: 52 mmol/mol
- eGFR: 82 mL/min/1.73m²
- Electrolytes: within normal limits
Ambulatory blood pressure monitoring confirms an average reading of 146/92 mmHg.
What is the most appropriate first-line management?
A. Lifestyle advice only
B. Start amlodipine
C. Start ramipril
D. Start bendroflumethiazide
E. Refer for urgent specialist review
Answers
Answer is C – Start ramipril.
This patient has stage 1 hypertension with a clinic blood pressure >140/90 mmHg and an ambulatory average >135/85 mmHg. He also has type 2 diabetes, which places him at higher cardiovascular risk. Current guidance recommends starting an ACE inhibitor as first-line treatment for hypertension in patients with diabetes, regardless of age.
Lifestyle advice alone would be insufficient given his comorbidity and confirmed diagnosis.
Amlodipine is first-line in patients over 55 years without diabetes, or in Black African or Caribbean patients.
Thiazide-like diuretics are typically considered second-line.
There are no features to suggest hypertensive emergency or secondary causes requiring urgent referral.



