The problem with weight loss drugs
Dear Friend,
I hope you’re finding your rhythm in 2026, however slow it feels. Starting the year with good intentions and honest reflection can be harder than it sounds, especially when the goal is something as stubborn and personal as weight loss.
This week I want to talk about something you’ve probably heard about everywhere: the new wave of weight-loss drugs — how they work, what the evidence shows, and what it really means for long-term health.
What Are These Drugs?
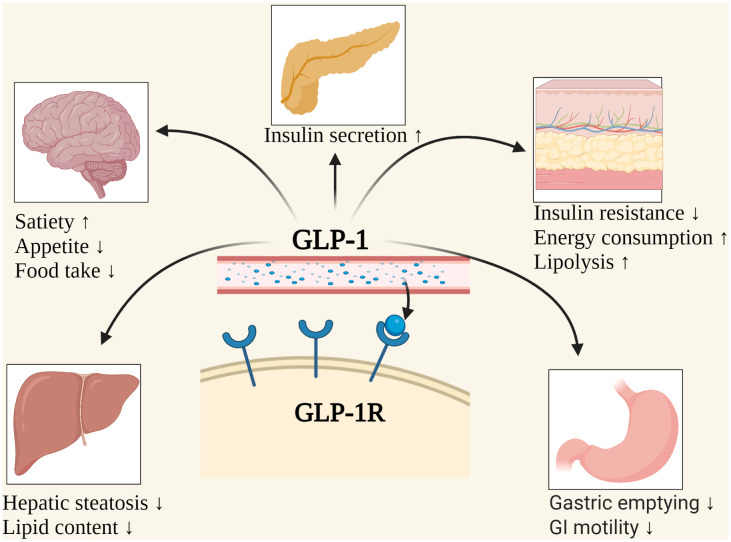
The medicines everyone’s talking about — like semaglutide and tirzepatide — belong to classes of drugs originally developed for type-2 diabetes treatment. They mimic or enhance hormones that help regulate appetite and metabolism. Specifically, they act on receptors for glucagon-like peptide-1 (GLP-1) — a hormone naturally released after eating that increases feelings of fullness, slows stomach emptying, and helps control blood sugar.
Tirzepatide also activates receptors for another hormone called GIP, which may further improve insulin use and energy balance. Together, these changes tend to reduce hunger, curb cravings, and help people eat less without constant hunger pangs.
Clinical trials have shown that, while on treatment, many people lose clinically meaningful amounts of weight — often more than with diet and exercise alone.
What Happens When People Stop the Drugs?
A new systematic review published in The BMJ — encompassing 37 studies and over 9,000 adults — sheds sobering light on what happens after stopping treatment. Those who stopped these medicines regained weight quickly — on average about 0.4 kg per month, returning to their original weight within roughly 1.5–2 years.
And crucially, the rate of weight regain after stopping medication was faster than after behavioural weight-loss programmes such as diet and exercise — almost four times faster by some estimates.
This was true even though people on drugs typically lost more weight in the first place. Improvements in markers like blood pressure, cholesterol and blood sugar returned toward pre-treatment levels once medication stopped.
So these drugs clearly work — and work well — while they are being taken. But the benefits don’t automatically persist once treatment ends.
A Chronic Condition, Not a Quick Fix
Obesity isn’t a short-lived problem that you zap once and it stays fixed. Biologically, the body strives to defend weight — appetite goes up and energy expenditure goes down after weight loss. These drugs blunt that drive while they’re in your system, but once they’re gone, the old signals come back.
That’s not a fault of the medication — it’s a reflection of the chronic, relapsing nature of obesity. The lesson from the evidence is simple but not sexy: long-term success usually requires long-term change — not just short-term pharmacology.
So What Actually Lasts?
This brings us back to basics we already know but often underplay:
Diet quality matters. Better nutrition fuels better metabolism, better energy, and better satiety without injections.
Movement matters. Exercise supports muscle mass, insulin sensitivity, mood, and appetite regulation.
Behaviour change matters. Learning how to eat slowly, observe hunger cues, manage stress, and build routines is the foundation of durable change.
Medication can be a powerful tool, especially for people with significant metabolic risk or disability from obesity. But if the only mechanism you rely on is a weekly jab, the evidence suggests that when you stop, much of what you gained slips away.
A Personal Note
I’ve seen patients and friends talk about these drugs as if they’re a magic “off switch” for appetite. They’re not. They’re better thought of as supportive medicine — something that buys you time and reduces biological hunger, while you build the skills that matter in the long run.
The goal isn’t perfection. It’s sustainable patterns you can carry forward — whether you’re on medication or not.
Drug of the week
Probenecid
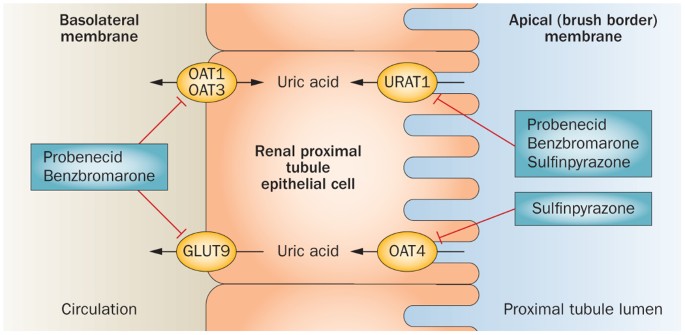
Probenecid is a uricosuric agent. It’s used mainly for chronic gout in patients who under-excrete uric acid.
It blocks renal tubular reabsorption of urate, increasing urinary uric acid excretion.
That same mechanism affects other organic acids.
It can raise levels of drugs like penicillin and some antivirals—sometimes deliberately.
Common side effects include gastrointestinal upset and rash.
The key risk is uric acid nephrolithiasis, especially without good hydration—one reason it’s used less often now.
A Brain Teaser
A patient is furious about an aspect of their care you provided. Before you’ve had a chance to discuss the situation they demand you full name and GMC reference number.
What should you do?
A: Withhold this information until you’ve had a chance to discuss the issue
B: Give them both details
C: Refuse to speak to them until she has calmed down
D: Withhold the information until you know what they intent to do with it
E: Instead give them information regarding your practice’s complaints procedure
Answers
The answer is B – give them both details.
The GMC states ‘If someone you have contact with in your professional role asks for your registered name and/or GMC reference number, you must give this information to them’
You cannot withhold this information.
GMC ethical guidance: Treat patients and colleagues fairly and without discrimination. Paragraph 64



